Cycloaddition reactions build ring-containing molecules by simultaneously creating two bonds between reacting molecules. Usually, two molecules with π-bonding combine to form a new cyclic molecule with reduced π-bonding. The most common cycloaddition reaction is the Diels-Alder reaction where a conjugated diene (2 π-bonds) reacts with an alkene (1 π-bond) to form a cyclohexene derivative (1 π-bond). Used appropriately, they provide a rapid means of generating molecular complexity in a controlled and predictable manner. For this reason, they are widely used in industry for the synthesis of medicines and agrochemicals.
Les réactions de cycloadditions génèrent des molécules cycliques en créant simultanément deux liaisons entre les substrats initiaux. Habituellement, deux molécules à liaisons π se combinent pour former une nouvelle molécule cyclique à liaison π réduite. La réaction de cycloaddition la plus courante est la réaction de Diels-Alder où un diène conjugué (2 liaisons π) réagit avec un alcène (1 liaison π) pour former un dérivé cyclohexénique (1 liaison π). Utilisées de façon appropriée, ces réactions fournissent un moyen rapide de générer une complexité moléculaire de manière contrôlée et prévisible. Pour cette raison, elles sont largement utilisées dans l’industrie pour la synthèse de médicaments et de produits agrochimiques.
Key Abstracts
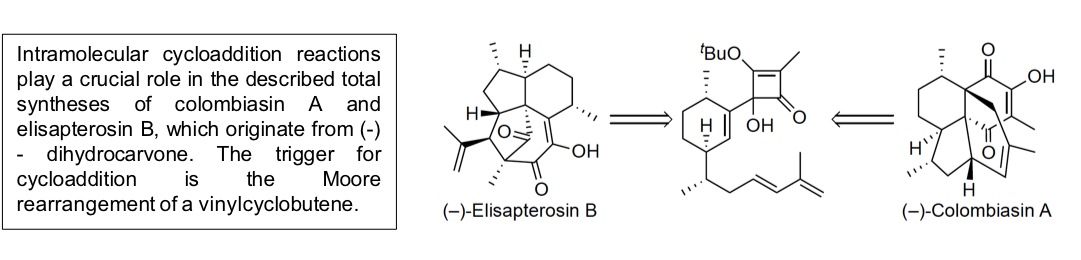
Total Synthesis of (-)-Colombiasin A and (-)-Elisapterosin B
D.C. Harrowven, D.D. Pascoe, D. Demurtas, H.O. Bourne, Angew. Chem, 2005, 117, 1247-1248
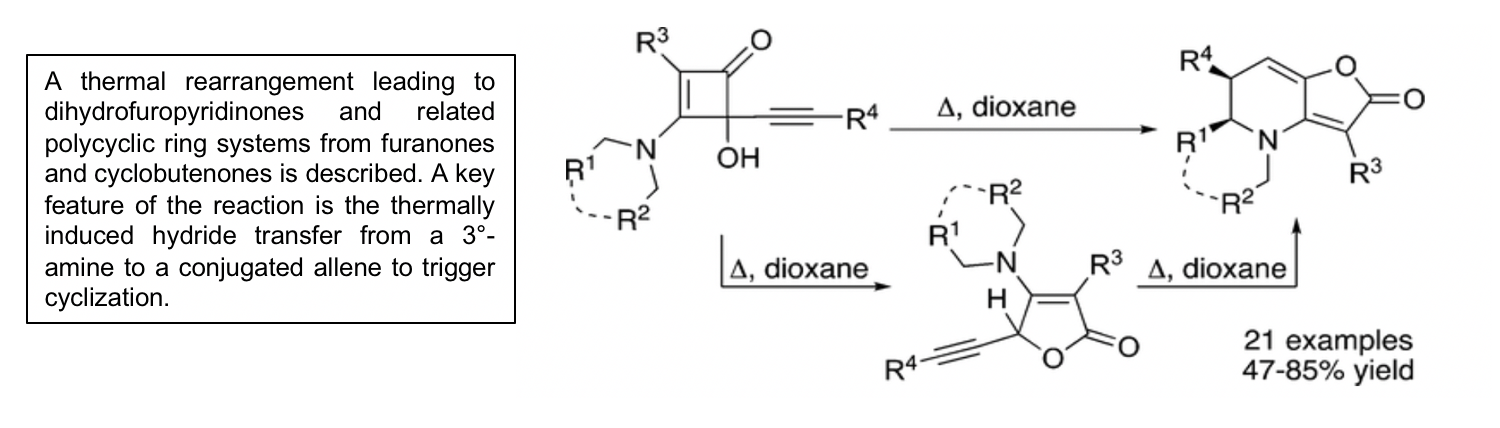
A Thermally Induced Hydride Transfer from an Amine to an Allene Triggers an Annulation Reaction, Giving Dihydrofuropyridinones
Members Involved




